

Cells sense nutrients such as amino acids (AAs) through the mammalian target of rapamycin complex 1 (mTORC1) signaling pathway to regulate growth and metabolism, which is a highly conserved survival adaptation in the process of biological evolution. In the tumor microenvironment (TME), despite widespread nutritional deficiencies, the scientific questions of why mTORC1 signaling in cancer cells is abnormally activated and how it is related to immune evasion during tumor progression have aroused great interest in the team.
After several years of research, the team found that various inflammatory factors in the TME (such as TNFα, IFNγ, etc.) "hijacked" the inherent amino acid sensing system of cancer cells by inducing the phosphorylation of the S127 residue of the ZBTB5 protein, driving abnormal activation of mTORC1, promoting tumor cells to absorb large amounts of amino acids, and triggering CD8+T cells undergo apoptosis due to nutrient starvation, leading to immune evasion, tumorigenesis, and immune drug resistance. During this process, ZBTB5-pS127 plays a key function as an inflammatory sensor. Specific blocking peptides are designed for this target, inhibiting tumor progression in various models, effectively reversing drug resistance, and greatly sensitizing immunotherapy.
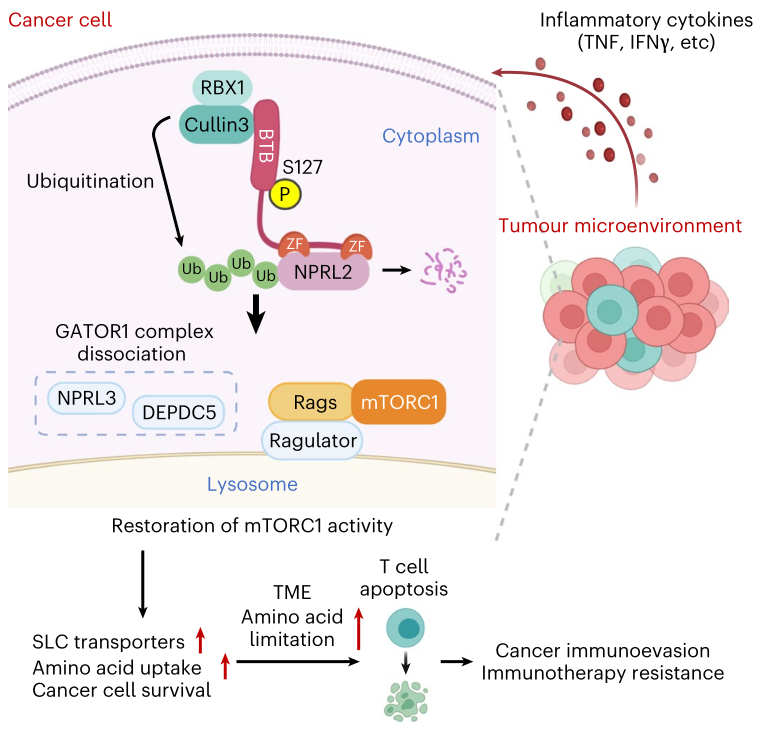 ▲Aberrant amino acid sensing promotes immunotherapy resistance through the inflammatory cytokine-ZBTB5-mTORC1 axis
▲Aberrant amino acid sensing promotes immunotherapy resistance through the inflammatory cytokine-ZBTB5-mTORC1 axis
The team analyzed various inflammation-related malignant tumors such as gastric cancer and found that the inflammatory state of the TME is closely related to the mTORC1 activity of cancer cells. In order to rigorously simulate the nutritional status of the TME, the team independently developed a culture system with a similar concentration of AAs in the tumor interstitial fluid, and found that the Ser127 site of the ZBTB5 protein (ZBTB5-pS127) was phosphorylated through JNK/p38 kinase when stimulated by inflammatory factors. This phosphorylation event promotes Cullin3 ZBTB5 The assembly of the E3 ubiquitin ligase complex specifically catalyzes the K48 polyubiquitination modification of NPRL2 protein and degrades it through the proteasome pathway, thereby destroying the integrity of the GATOR1 complex. Since the GATOR1 complex is a key inhibitor of the mTORC1 nutrient sensing signaling pathway, cancer cells maintain high activation of mTORC1 in the nutrient-starved TME through the above method. Therefore, this novel cell signaling pathway represents the core molecular pathway by which inflammatory signals “hijack” nutrient sensing pathways.
In terms of tumor biological functions, the team found that the ZBTB5-pS127-mTORC1 axis has a small effect on cancer cell proliferation. It mainly increases the uptake of amino acids (including methionine, glutamine, etc.) by cancer cells by upregulating a variety of SLC family transport proteins (such as SLC6A14, SLC43A2), thereby significantly reducing amino acid levels in tumor interstitial fluid. This nutritional deprivation causes the infiltrated CD8⁺T cells to undergo apoptosis due to "starvation", thereby inhibiting anti-tumor immunity and achieving immune escape. In a variety of immunotherapy clinical tumor samples, activation of this signaling axis is closely associated with reduced CD8⁺T cell infiltration, poor response to anti-PD-1 therapy, and poor prognosis. In terms of targeted intervention, blocking the ZBTB5-pS127 site can specifically block Cullin3 ZBTB5 The complex forms, upregulates TME amino acid levels, restores CD8⁺T cell activity, significantly inhibits tumor growth, prolongs survival, and reverses primary and acquired resistance to immunotherapy in a variety of mouse models. These findings bring new opportunities for improving tumor immunotherapy, especially for drug-resistant patients.
"Nature Cell Biology" is sponsored by Nature Portfolio. It publishes research, reviews and comments on cell biology and related fields that have major scientific breakthroughs and application potential, providing cutting-edge academic references and mechanistic insights for cell biologists, molecular biologists and medical researchers. The impact factor in 2024 is 19.1, JCR partition Q1.
Click below to read the original text and access the original text link.