


Colorectal cancer (CRC) is one of the most common malignant tumors in the world. At present, traditional strategies based on surgery, radiotherapy, chemotherapy, and combination therapy have limited efficacy for metastatic patients, and emerging immunotherapies can only benefit a small number of patients. Therefore, exploring new treatment targets and improving patient prognosis have become key issues that need to be solved urgently in the field. The occurrence and development of CRC involves multiple levels such as gene mutation accumulation, metabolic reprogramming, epigenetic changes, and tumor microenvironment remodeling. Among them, metabolic reprogramming, as an important feature of solid tumors, has shown great potential in terms of clinical relevance and targeted drug development. In particular, lipid metabolism reprogramming has attracted much attention. However, as a synthetic scaffold for various lipids, the role of metabolic reprogramming of fatty acids in the progression of CRC is unclear. Whether it affects the tumor microenvironment and the specific mechanism have not yet been elucidated.
The research team first analyzed multiple sets of transcriptome sequencing data and combined with clinical information to screen out the key enzyme - ACADS - that is closely related to fatty acid reprogramming in CRC. Subsequently, various CRC models were constructed in immune-competent mice, nude mice, and conditional knockout mice, and combined with single-cell sequencing, multiplex immunofluorescence, and flow cytometry techniques, it was confirmed that ACADS exerts its tumor suppressive function by reshaping the tumor immune microenvironment. In order to further clarify the mechanism, the team used sequencing and intervention experiments at the cell and animal levels to reveal that the mtDNA-mediated cGAS-STING signaling pathway is a key link in the anti-tumor effect of ACADS. After in-depth investigation, it was found that the reduction in mtDNA omission caused by the deletion of ACADS was closely related to its own modification. With the help of mass spectrometry analysis, co-immunoprecipitation and super-resolution microscopy, the study confirmed that ACADS interacts with mito-DNMT1 and affects the methylation level of mtDNA. Interfering with the expression of mito-DNMT1 can reproduce the ACADS-related phenotypes, further verifying the key role of this regulatory axis.
To verify the clinical relevance of the ACADS-mito-DNMT1-cGAS-STING signaling pathway, the team collected samples from 77 colorectal cancer patients for analysis. The results showed that this signaling pathway is closely related to the malignant progression of CRC. Finally, the team identified hypericin as an activator of ACADS through virtual screening. Functional experiments showed that the anti-tumor effect of hypericin is ACADS-dependent, further confirming the feasibility of ACADS as a potential therapeutic target.
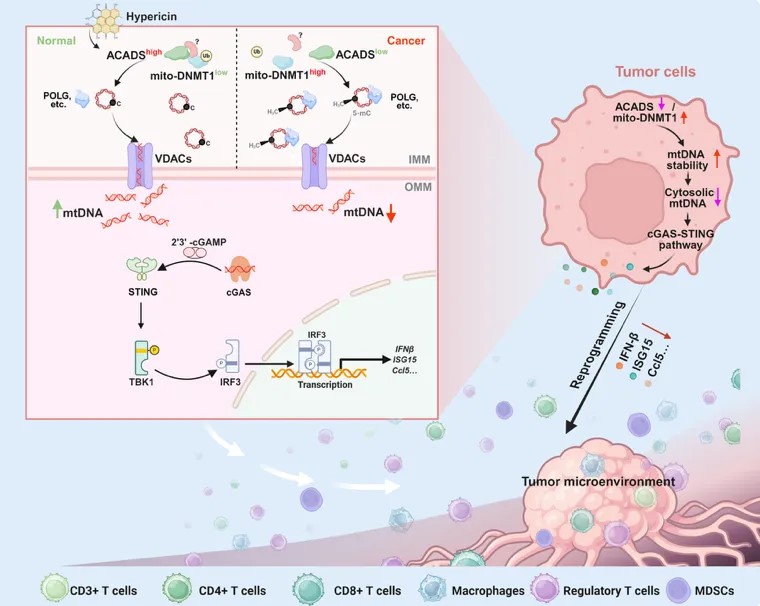
▲ACADS activates anti-tumor immune mechanisms by regulating mitochondrial DNA methylation
In summary, this study revealed a new mechanism by which fatty acid metabolism reprogramming promotes immune evasion in colorectal cancer, and screened out the natural small molecule compound hypericin that targets and activates the ACADS-mito-DNMT1-cGAS-STING pathway. It is worth noting that hypericin has entered phase III clinical trials in other tumors and has a good basis for clinical translation. This discovery provides a strong theoretical basis and potential drug candidate for the ACADS-based targeted treatment strategy for colorectal cancer, which has important scientific significance and clinical prospects.
"Signal Transduction and Targeted Therapy" is published by Springer Nature and publishes original research articles and review articles related to various aspects of signal transduction in physiological and pathological processes, as well as signal transduction-targeted therapies in the form of biological agents and small molecule drugs for the treatment of human diseases (such as cancer, autoimmune diseases, etc.). The impact factor in 2025 is 52.7, and the JCR division is Q1.
Click below to read the original text and access the original text link.