

Immune checkpoint inhibitors represented by PD-1/PD-L1 have significantly changed the treatment landscape of malignant tumors, but their clinical application still faces key issues such as limited response rates, frequent immune-related adverse reactions, and complex drug resistance mechanisms. PD-L1 expression on myeloid cells in the tumor microenvironment is a core node in regulating anti-tumor T cell immune responses. However, the upstream secreted regulators, specific receptors and downstream signaling pathways that drive PD-L1 expression in myeloid cells in the tumor microenvironment have not yet been systematically elucidated. Human epididymis protein 4 (HE4) is a secreted glycoprotein that is abnormally highly expressed in various tumors such as lung adenocarcinoma and ovarian cancer. As a commonly used clinical tumor marker, its role and molecular mechanism in tumor immune microenvironment remodeling and immune escape are still unclear. Based on this, this study aims to reveal the specific mechanism of HE4 regulating PD-L1 in myeloid cells, clarify its function in tumor immune escape, and explore its translational value as an immunotherapy target and efficacy prediction marker.
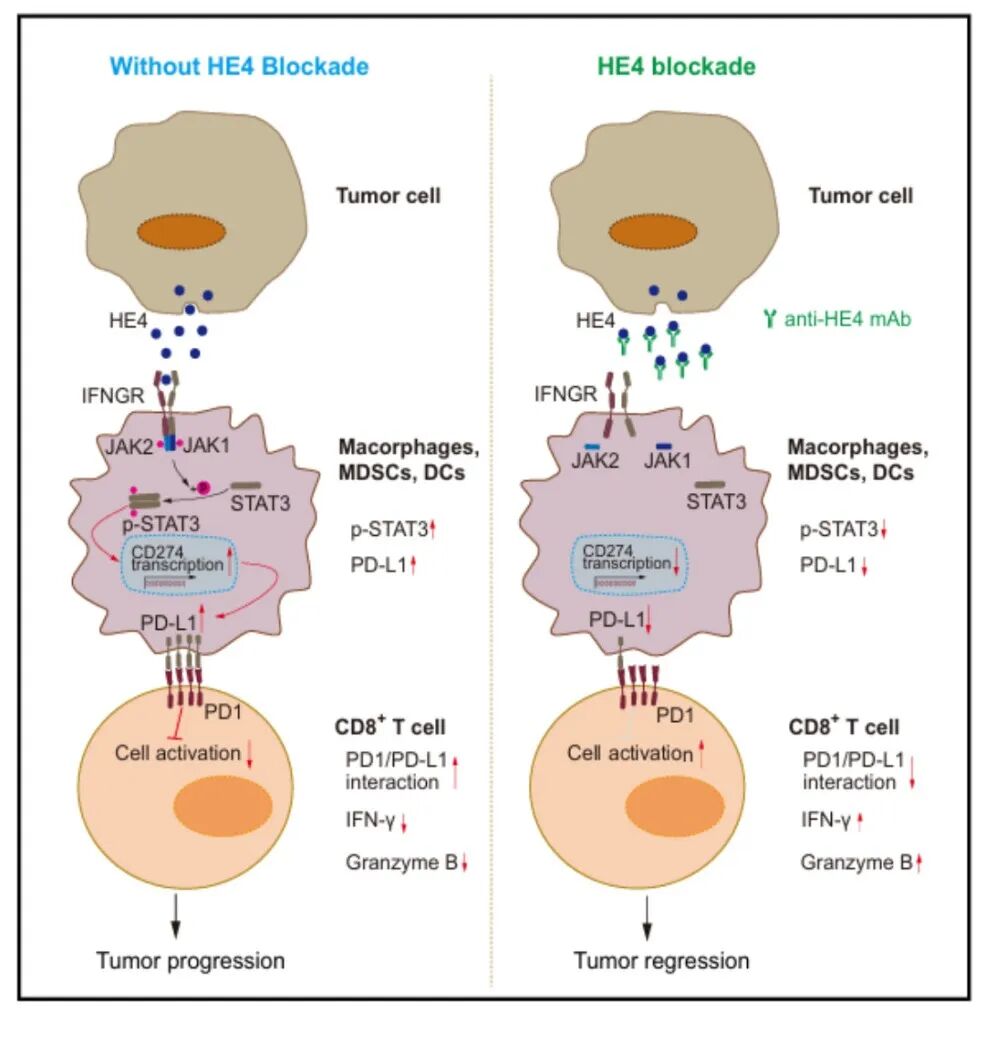 ▲The mechanism of HE4 regulating myeloid PD-L1-mediated tumor immune evasion
▲The mechanism of HE4 regulating myeloid PD-L1-mediated tumor immune evasion
This study carried out systematic research through clinical sample analysis, animal models, molecular biochemical experiments and single cell sequencing. The results show that HE4 is significantly highly expressed in lung adenocarcinoma and ovarian cancer tumor tissues, and its role in promoting tumor progression does not rely on direct regulation of tumor cells, but is achieved through the adaptive immune pathway. Mechanistically, HE4 secreted by tumors can directly bind to the IFN-γR1/2 receptors on the surface of myeloid cells, selectively activate the JAK-STAT3 signaling pathway, and upregulate PD-L1 expression at the transcriptional level. This process does not rely on classic pathways such as IFN-γ, STAT1 and NF-κB. ; At the same time, the concentration of HE4 in the tumor microenvironment is much higher than that of IFN-γ. It can competitively bind to IFN-γR through concentration advantage, inhibit the activation of the IFN-γ-STAT1 pathway, reduce its pro-inflammatory and anti-tumor effects, and reprogram myeloid cells to transform into an immunosuppressive phenotype. Functional experiments have confirmed that HE4 knockout or neutralizing antibody blockade can significantly reduce PD-L1 levels in macrophages, dendritic cells and myeloid-derived suppressor cells, and restore CD8 +The activation and effector functions of T cells can effectively inhibit tumor growth in various mouse models such as lung cancer and ovarian cancer, and can produce synergistic effects with anti-CTLA-4 immunotherapy and paclitaxel chemotherapy. Safety evaluation shows that the incidence of inflammatory factor release, multi-organ immune inflammation, and liver and kidney function damage caused by anti-HE4 treatment is significantly lower than that of PD-1 inhibitors, and has a better therapeutic safety window. Clinical cohort validation shows that high HE4 expression in lung adenocarcinoma tissue is significantly associated with good response to PD-1 inhibitor treatment, and its predictive performance is better than traditional PD-L1 markers.
This study reveals for the first time that HE4-IFN-γR-JAK-STAT3-PD-L1 is a new molecular axis that regulates myeloid immune suppression and tumor immune escape. It clarifies the key mechanism by which HE4, as a tumor-derived secreted protein, regulates the tumor immune microenvironment, and improves the transcriptional regulatory network of PD-L1. The research results confirm that HE4 has the dual value of both a tumor immunotherapy target and a PD-1/PD-L1 efficacy prediction marker, providing a new theoretical basis and important translation direction for the development of safer and more efficient tumor precision immunotherapy strategies.
"Cell Reports Medicine" is published by Cell Press and publishes cutting-edge translational and clinical biomedical research to provide information and impact on human health and medicine.
Click below to read the original text and access the original text link.