
Recently, Professor Zhang Jianxiang's team of the Army Military Medical University and the Chongqing-Guangdong Pathological Science Research Center and Professor Russell New Team of the First Affiliated Hospital of Chongqing Medical University worked together in international journalsAdvanced Functional MaterialsA research article "Bifunctional Peptide-Engineered Mitochondria Alleviate Myocarditis and Cardiac Fibroosis by Site-Specifically Inhibiting NETosis" was published on the study. The study found that mitochondrial transplantation can inhibit NETosis by regulating the mitochondrial function of neutrophils and engineer the mitochondria to further improve the therapeutic effect. This work provides a new strategy for the development of innovative mitochondrial-derived therapies for myocarditis and other autoimmune and inflammatory diseases.

As the main cause of sudden cardiac death and dilated cardiomyopathy in young and middle-aged people, myocarditis mainly relies on glucocorticoid treatment in clinical practice, and has certain limitations. Effective treatment methods are urgently needed at present. This study found that in experimental autoimmune myocarditis (EAM) mouse model, exogenous mitochondria (Mito) transplantation significantly reduces cardiac inflammation and oxidative stress, improves myocardial injury, improves cardiac function, and relieves fibrosis and heart failure. The mechanism of action is: the injected mitochondria are internalized by neutrophils and transported to the inflammatory heart. By inducing mitochondrial fusion, restoring mitochondrial function, inhibiting the opening of mitochondrial permeability conversion pores (mPTP) and reducing the excessive generation of mitochondrial reactive oxygen species (mtROS), jointly inhibiting the formation of neutrophil extracellular trapping nets (NETs) (i.e. NETosis), thereby blocking NETs-mediated cardiomyocyte inflammation and apoptosis. It is worth noting that this study reveals for the first time that mitochondrial transplantation may play a therapeutic role by regulating neutrophils rather than directly integrating into bull’s myocardial cells. The further developed bifunctional peptide (SDKP-SS31) engineered mitochondrial combination therapy (with anti-inflammatory and anti-fibrotic properties) has shown significant synergistic efficacy in mouse models, providing innovative mitochondria-derived treatment for myocarditis and other autoimmune/inflammatory diseases.
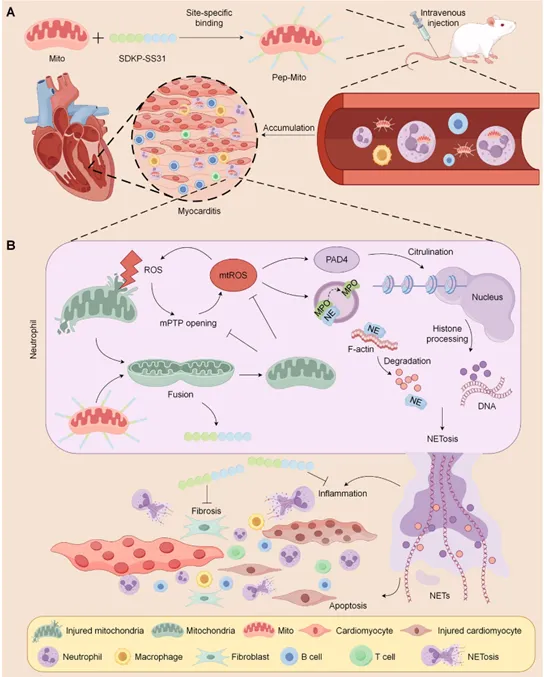
Figure 1: Schematic diagram of bifunctional peptide-engineered mitochondria in the treatment of myocarditis mice.
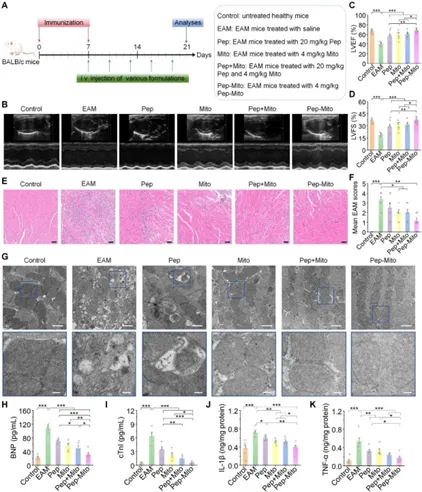
Figure 2: Effects of bifunctional peptide-engineered mitochondria in the treatment of EAM mice (Day 21).
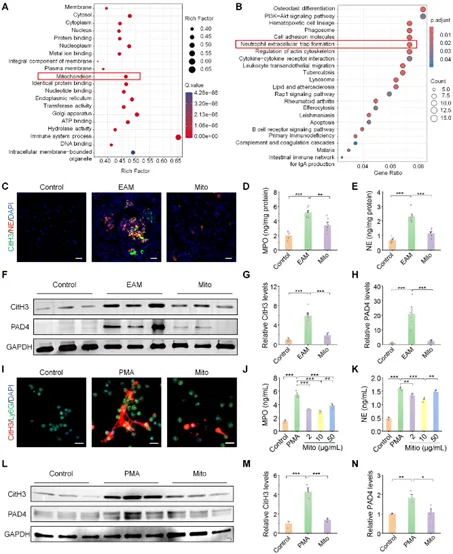
Figure 3: Exogenous mitochondria (Mito) inhibits the formation of neutrophil extracellular trapping nets (NETs) in vivo and in vivo.
Advanced Functional Materials is a top journal in the field of materials science (influence factor 19.0, JCR Q1 area and the TOP journal of Materials Science Major Area 1, Chinese Academy of Sciences). It was published by Wiley-VCH Verlag Publishing House and was founded in 2001. The journal focuses on interdisciplinary innovations such as nanotechnology, energy materials, and biomedical engineering. It prioritizes research with disruptive functional breakthroughs and clear application scenarios. It is an authoritative platform for reporting cutting-edge achievements in the field of materials.
Click on the lower left to read the original text and access the original text link.
https://pubs.acs.org/doi/10.1021/acsnano.5c02492