
Recently, Professor Zhu Bo’s team from Xinqiao Hospital of the Army Medical University and Jinfeng Laboratory, worked with Professor Li Qijing of Singapore Science and Technology Research Bureau (A*STAR) and Professor Su Chunxia of Shanghai Pulmonary Hospital to publish a research paper titled “Bone metastases diminish extraosseous response to checkpoint blockade immunotherapy through osteopontin-producing osteoclasts” in the international journal Cancer Cell.

The emergence of immune checkpoint inhibitors (ICBs) brings long-term survival hopes for patients with advanced cancer, but the low clinical response rate remains a serious challenge for their clinical application. Among them, ICB efficacy in patients with bone metastasis is particularly poor, and the overall survival time is significantly shortened. Although bone metastasis can have a incidence of up to 70% in high incidence cancers such as breast cancer, prostate cancer, and lung cancer, it leads to long-term unknown mechanisms of immunotherapy resistance, and lacks targeted intervention strategies. This clinical "blind spot" forces patients to fall into the dilemma of "hard to be effective with medicine".
Professor Zhu Bo’s team conducted a multi-center retrospective analysis of more than 2,000 patients with metastatic tumors (including 11 types of cancers such as lung cancer and breast cancer), revealing for the first time a key clinical phenomenon: the presence of bone metastasis will specifically weaken the response of patients’ extra-brain lesions (such as lung, liver, and lymph nodes) to ICB immunotherapy. After excluding confounding factors such as tumor burden, the objective response rate of extra-brain lesions in patients with bone metastasis decreased by 10.8%, and the risk of disease progression increased by 57% to 123%. Imaging analysis showed that after patients with bone metastasis received ICB, extra-bone tumors had almost no withdrawal (average reduction ratio <5%), while the group without bone metastasis decreased significantly.
To reveal the immunologic mechanism behind this phenomenon, researchers innovatively constructed a dual tumor model: implanting bone metastasis foci in the tibia of mice (simulating clinical bone metastasis) and establishing a second tumor subcutaneously or in the lungs (simulating extra-bone lesions). When PD-L1 inhibitor was administered: mice with subcutaneous tumor-bearing alone responded significantly ; When bone metastasis was accompanied, the extra-bone lesions (subcutaneous/pulmonary metastasis lesions) in the same mouse were almost unresponsive and T-cell infiltration was reduced. Through serum reentry experiments, the blood of bone metastasized mice carries "immunosuppressive factors" and can reduce the therapeutic response of boneless metastasized mice to ICB. This result reproduces clinical observations and lays the foundation for mechanism exploration.
Through quantitative proteomic screening and analysis mining, researchers focused on osteoclasts, the core role in the bone microenvironment. Bone metastasis remodels osteoclast function, causing it to secrete osteopontin (OPN): The serum OPN level of bone metastasis mice is increased by about 2 times, and 70% are derived from osteoclasts. OPN reaches extra-skeletal tumors through blood circulation, inhibiting the differentiation of the key subpopulation Tpex (stem cell-like exhausted precursor cells) in CD8+ T cells, a core cell population in response to ICB treatment. Gene knockout experiments confirmed that after specifically clearing the OPN of osteoclasts, the ICB treatment response of extra-bone tumors was significantly restored, and the number of Tpex cells returned to normal levels.
Based on mechanism research, the team proposed a new strategy to overcome resistance to bone metastasis: targeting the upstream activation signal of osteoclasts RANKL. The study included the commonly used bone protection drug disumab (a neutralizing antibody for RANKL). In a cohort containing 152 patients with bone metastasis, the extra-bone lesions in the ICB combined with desusumab treatment group were significantly reduced, and the efficacy was significantly better than that of the ICB alone or the ICB combined with traditional bisphosphonate treatment group. The median progression-free survival (PFS) in the combined treatment group was extended to 357 days, significantly better than the 167 days in the control group (hazard ratio HR=0.47, p<0.001). Typical cases show that after a patient with drug-resistant nasopharyngeal carcinoma had reduced extra-brain lesions by more than 50% after switching to ICB combined with desulsumab. It also supports the inhibition of osteoclast activity and reversible immunotherapy resistance. The study also established a clinical prediction system: serum OPN level can predict the efficacy of ICB-PFS shortening in patients with high OPN levels at baseline ; Patients whose OPN has not decreased during treatment have an increased risk of tumor progression. To sum up, the existing clinical drug disuzumab is expected to solve the dilemma of ICB resistance in extra-bone lesions caused by bone metastasis, and provide patients with a practical solution.
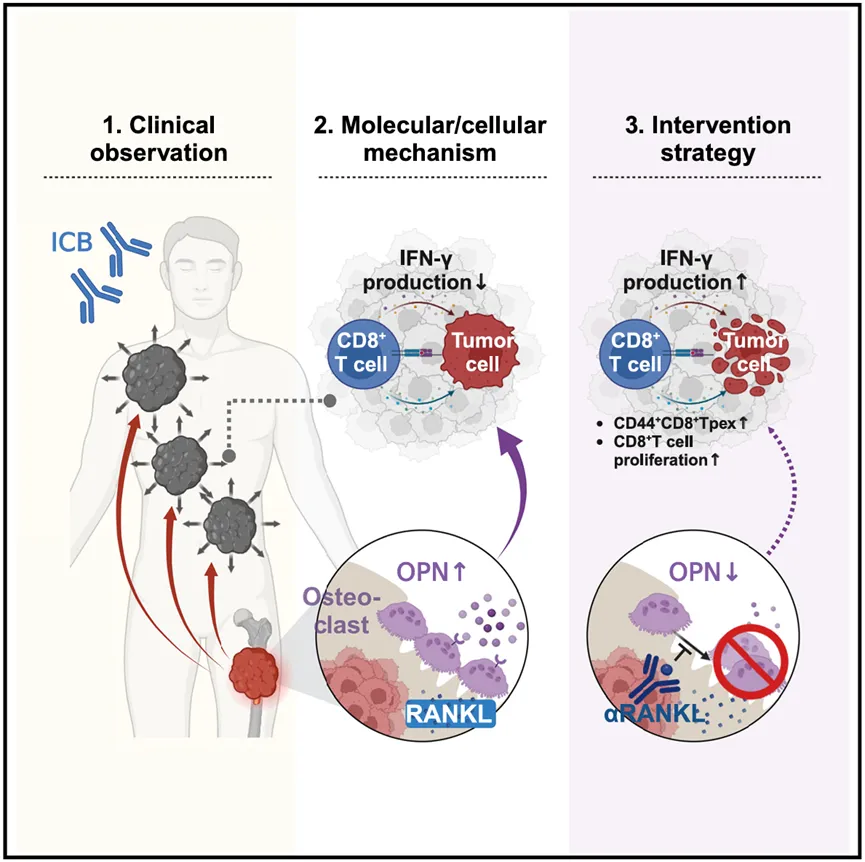
▲Schematic diagram of the mechanism of bone metastasis leading to tumor immunotherapy resistance.
Cancer Cell is a top journal in the international oncology field (impact factor 44.5, JCR Q1 area). It was founded by Cell Press in 2002 and has a monthly publication cycle. The journal focuses on publishing major breakthrough results in cancer biology, tumor mechanisms and clinical translational research, emphasizing in-depth exploration of important scientific issues of naturally occurring cancer, and giving priority to publishing clinical studies that can promote the establishment of new paradigms for cancer treatment, diagnosis or prevention. It has long been ranked in the TOP journal of the Cell Biology and Oncology subcategory, and is an authoritative platform for scientific researchers in this field to publish cutting-edge results.
Click on the lower left to read the original text and access the original text link.
https://pubs.acs.org/doi/10.1021/acsnano.5c02492